

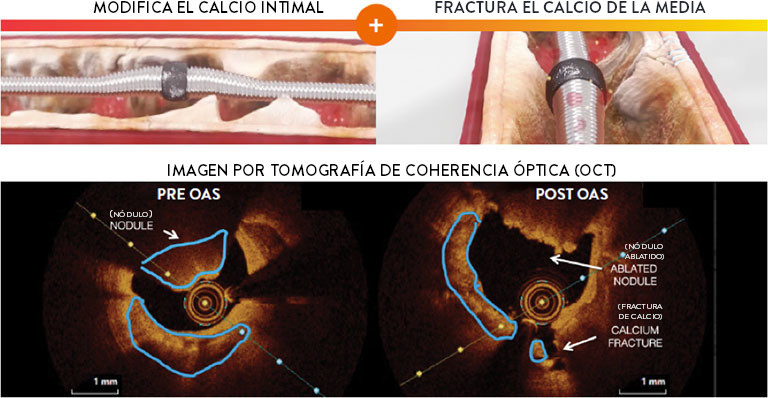
Diamondback 360™ es el único sistema de aterectomía con una corona revestida de diamante que orbita 360 grados, diseñado para abordar el calcio severo.
El sistema de aterectomía orbital coronaria (OAS) del Diamondback 360™ utiliza un mecanismo de acción dual único, que combina lijado diferencial y fuerzas pulsátiles, permitiendo la modificación simultánea del calcio intimal y medial para la entrega, expansión y aposición ideales del stent en lesiones severamente calcificadas. Un solo dispositivo trata calcio excéntrico, concéntrico y nodular.1-6
Diamondback 360™ OAS tiene un Diseño Único con Mecanismo de Acción Dual
Un único dispositivo reduce el calcio de la intima y fractura el calcio de la media. El lijado diferencial y las fuerzas pulsátiles permiten la modificación simultánea tanto del calcio de la intima y de la media para la entrega, expansión y aposición óptimas del stent en lesiones severamente calcificadas1-4

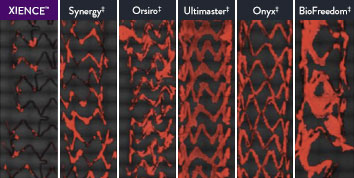
The XIENCE™ Stent is also recognized as being significantly more anti-thrombotic than other DES on the market. As shown in the study findings, XIENCE™ Stent reveals significantly less (p < 0.01) platelet adhesion—shown in red in the confocal microscopy images—than other DES, and platelet adhesion is an important factor in stent thrombosis.*8 These findings suggest that this stent choice “may be ideally suited for very short-term DAPT.”8
*Ex Vivo Swine Shunt Model.

El Diamondback 360™ OAS ofrece la versatilidad necesaria para tratar casos desafiantes
Trate incluso las lesiones más severamente calcificadas, con configuración en menos de 2 minutos7,8 y tiempos de procedimiento predecibles.2
Facilita el Tratamiento Anterógrado y Retrógrado de:
- Lesiones Largas y difusas
Tratamiento exitoso de lesiones de hasta 60 mm de longitud en estudio real.9 - Lesiones Severamente Estenóticas
Cruce de más del 99% de las lesiones con menos del 2% de pre-dilatación en el estudio ORBIT II.1,10 - Lesiones Nodulares
Tratamiento eficaz de la calcificación nodular.5,6 - Lesiones Ostiales
Tratamiento seguro de lesiones ostiales.11-13
Perfil Bajo
Compatible con 6F para acceso femoral o radial.14
Varios Tamaños de Vasos
Una sola corona de 1,25 mm trata vasos de 2,5 mm a 4,0 mm14
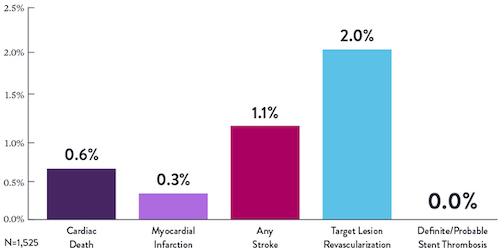
Seguridad Comprobada |
Éxito del Procedimiento |
Baja Tasa de
|
|
>2,200 |
<1% |
97.7% |
0.9% |
|
Pacientes en 11 |
Complicaciones Angiográficas |
Cruce e implantación del stent |
En el Estudio |
Desempeño Clínico Sostenido
Los datos son para TLR en el estudio ORBIT II en la cohorte de pacientes OA+DES.


“Stopping DAPT at 3 months in selected patients after [XIENCE™ Stent] implantation was at least as safe as the prolonged DAPT regimen adopted in the historical control group.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT 2 Trial Design and Randomization10

Short 1-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- After 1-month: Clopidogrel monotherapy

12-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- 1 to 12-month: Aspirin + Clopidogrel
- 12 to 60-month: Aspirin monotherapy

- Successful PCI using CoCr everolimus-eluting stent: XIENCE™
- Eligible for DAPT (aspirin/P2Y12 receptor blocker) for 1 year

- Patients who need oral anticoagulants
- History of intracranial bleeding
- Major in-hospital complications (MI/stroke/major bleeding)
STOPDAPT Study: XIENCE™ Stent with 3-Month DAPT Is Feasible9
STOPDAPT9 was the first prospective trial to study DAPT cessation at 3 months after implantation. Among other 1-year outcomes, the XIENCE™ Stent rate of stent thrombosis was 0.0%.
STOPDAPT Study Demonstrates Feasibility of XIENCE™ Stent with 3-Month DAPT9

Learn more about STOPDAPT 2

“It was noteworthy that no definite or probable stent thrombosis occurred in [XIENCE™ Stent] patients enrolled in STOPDAPT.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT-3 Trial Design and Randomization11

- PCI with planned exclusive use of CoCr-EES (XIENCE)
- ACS presentation or ARC-HBR
- Eligible for DAPT (Aspirin/P2Y12inhibitor) for 1 month
Study design and Randomization
Group 1:
0 to 1-month: Aspirin + P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
Group 2:
0 to 1-month: P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
STOPDAPT-3 Trial 11 was designed to explore 0-month DAPT* (SAPT˄ using only P2Y12 inhibitor) for ACS and HBR patients.
Though the results are comparable for both bleeding and ischemic events for DAPT and SAPT arms, the study did not meet its endpoint and concluded to use DAPT for 1 month after PCI.
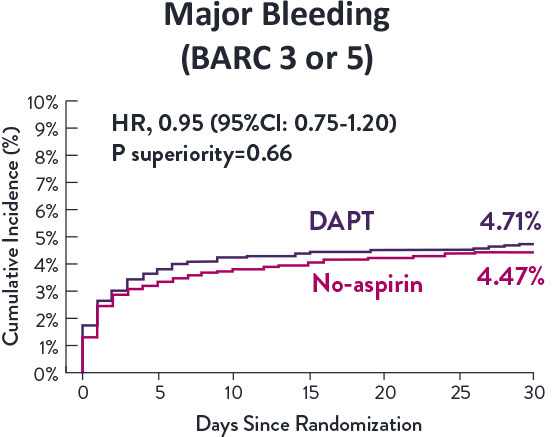
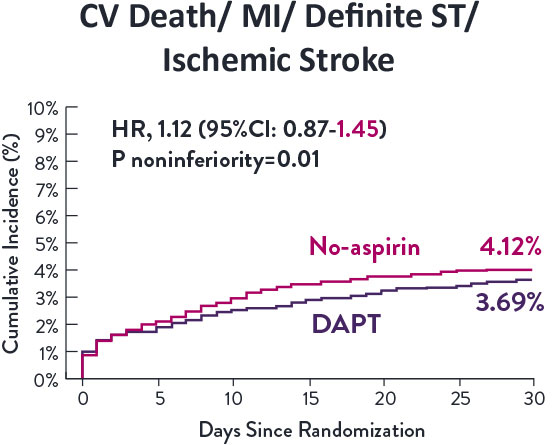

XIENCE™ Stent remains the ONLY DES with the shortest DAPT indication, as short as 28 days.12


Referencias
- Chambers, J., et al., JACC Cardiovasc Interv. 2014;7(5):510-518.
- Shlofmitz, E., et al., Expert Rev Med Devices. 2017;14(11):867-879.
- Yamamoto, M., et al., Catheter Cardiovasc Interv. 2019;93(7):1211-1218.
- Kini, A., et al., Catheter Cardiovasc Interv. 2015;86(6):1024-1032.
- Shlofmitz, E., et al., Interv Cardiol. 2019;14(3):169-173.
- Abellas-Sequeiros, M., et al., REC Interv Cardiol. 2022;4(2):163-164.
- Lee, MS, et al., J Invasive Cardiol. 2016;28(4):147-150.
- Moses, J., Cardiac Interventions Today. 2015;May/June:75. Link accessed November 20, 2023. https://citoday.com/articles/2015-may-june/vesselpreparation-with-orbital-atherectomy-for-treating-severe-coronary-calcium-for-stent-delivery.
- Vinardell J, et al. J Am Coll Cardiol. 2020;76(17 Suppl S):B71.
- Data on file at Abbott. In the ORBIT II study, the OAS was inserted and activated in 434 subjects, but in 2 cases, the OAS was unable to cross the lesion.
- Lee MS, et al. J Interv Cardiol. 2018;31(1):15-20.
- Chambers JW, et al. Catheter Cardiovasc Interv. 2022;100(4):553-559.
- Ghazzal A, et al. Cardiovasc Revasc Med. 2024;58:52-57.
- Diamondback 360™ Coronary Orbital Atherectomy Systems Instructions for Use (IFU). Please refer to IFU for additional information.
- Data on file at Abbott.
- Lee, MS, et al., J Interv Cardiol. 2016;29(4):357-362.

MAT-XXXXXXX



