

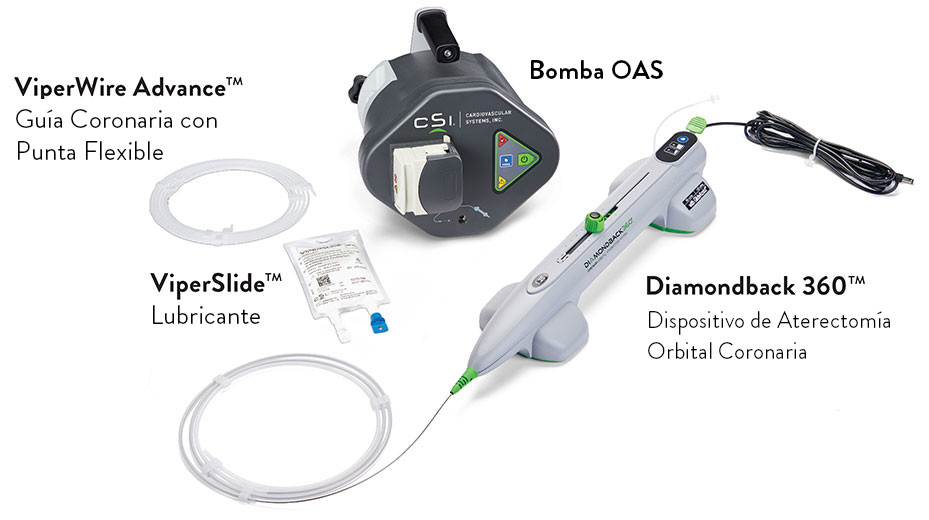
Sistema de Aterectomía Orbital Coronaria Diamondback 360™1
El Diamondback 360™ es el único sistema de aterectomía con una corona revestida de diamante que orbita 360 grados, diseñado específicamente para abordar el calcio severo para la optimización de la entrega, expansión y aposición del stent.2-5

![]() Compatible con Guía 6 Fr
Compatible con Guía 6 Fr
![]() Cable Eléctrico
Cable Eléctrico
![]() Botón de Inicio con un toque facilita el inicio rápido y sin esfuerzo del dispositivo
Botón de Inicio con un toque facilita el inicio rápido y sin esfuerzo del dispositivo
![]() Dos controles de velocidad convenientes permiten ajustes rápidos dentro del campo estéril
Dos controles de velocidad convenientes permiten ajustes rápidos dentro del campo estéril
![]() Botón de presurización permite el lavado rápido dentro del campo estéril
Botón de presurización permite el lavado rápido dentro del campo estéril
Corona Única de 1,25 mm1

Componentes del sistema
Configuración del sistema en menos de 2 minutos6,7
Modo GlideAssist™
El GlideAssist™ permite que la corona gire a una velocidad lenta (5K RPM), reduciendo la fricción en el sistema para:8

- Facilitar el seguimiento del dispositivo
- Facilitar la extracción del dispositivo
- Permitir un reposicionamiento más suave del ViperWire Advance™ durante los procedimientos

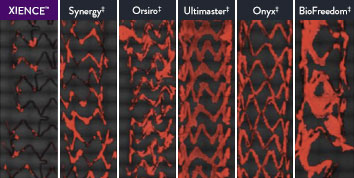
The XIENCE™ Stent is also recognized as being significantly more anti-thrombotic than other DES on the market. As shown in the study findings, XIENCE™ Stent reveals significantly less (p < 0.01) platelet adhesion—shown in red in the confocal microscopy images—than other DES, and platelet adhesion is an important factor in stent thrombosis.*8 These findings suggest that this stent choice “may be ideally suited for very short-term DAPT.”8
*Ex Vivo Swine Shunt Model.

Diamondback 360™ OAS Gives You the Versatility to Treat Challenging Cases
Treat even the most severely calcified lesions, with under 2-minute setup7,8 and predictable procedure times.2
Facilitates antegrade and retrograde treatment of:
- Long, Diffuse Lesions
Successfully treated lesions up to 60 mm in length in real-world study.9 - Heavily Stenosed Lesions
Crossed >99% of lesions with <2% pre-dilatation in the ORBIT II study.1,10 - Nodular Lesions
Effectively treats nodular calcification.5,6 - Ostial Lesions
Safely treats ostial lesions.11-13
Low Profile
6F compatible for femoral or radial access.14
Multiple Vessel Sizes
A single 1.25 mm crown treats vessels 2.5 mm to 4.0mm14
|
|
Proven Safety |
Procedural Success |
Low Q-Wave MI Rate |
>2,200 |
<1% |
97.7% |
0.9% |
|
Patients Across 11 |
Component Angiographic |
Crossing and Stent |
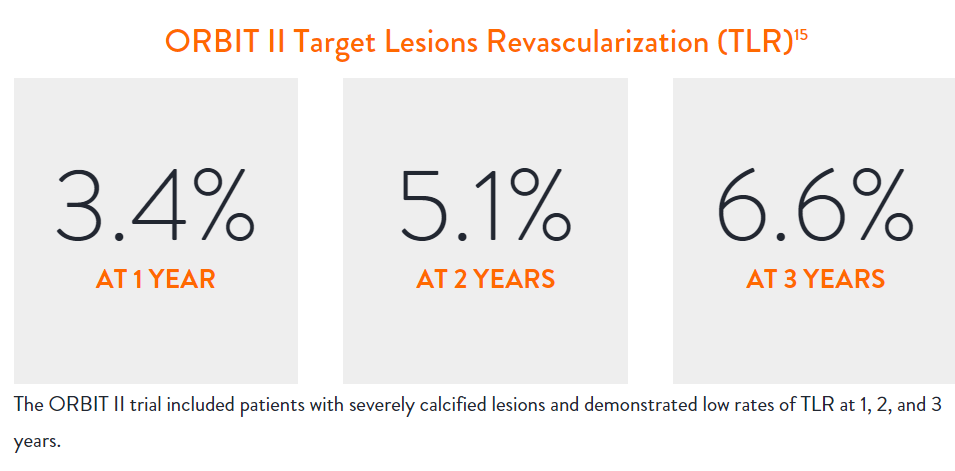
In the ORBIT II |
Sustained Clinical Performance
Data are for ORBIT II TLR in the OA+DES patient cohort.

“Stopping DAPT at 3 months in selected patients after [XIENCE™ Stent] implantation was at least as safe as the prolonged DAPT regimen adopted in the historical control group.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT 2 Trial Design and Randomization10

Short 1-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- After 1-month: Clopidogrel monotherapy

12-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- 1 to 12-month: Aspirin + Clopidogrel
- 12 to 60-month: Aspirin monotherapy

- Successful PCI using CoCr everolimus-eluting stent: XIENCE™
- Eligible for DAPT (aspirin/P2Y12 receptor blocker) for 1 year

- Patients who need oral anticoagulants
- History of intracranial bleeding
- Major in-hospital complications (MI/stroke/major bleeding)
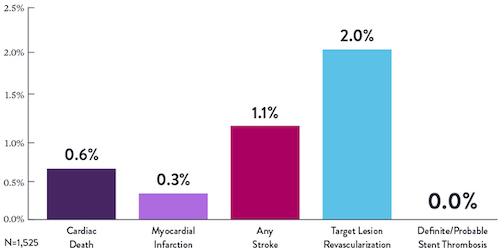
STOPDAPT Study: XIENCE™ Stent with 3-Month DAPT Is Feasible9
STOPDAPT9 was the first prospective trial to study DAPT cessation at 3 months after implantation. Among other 1-year outcomes, the XIENCE™ Stent rate of stent thrombosis was 0.0%.
STOPDAPT Study Demonstrates Feasibility of XIENCE™ Stent with 3-Month DAPT9

Learn more about STOPDAPT 2

“It was noteworthy that no definite or probable stent thrombosis occurred in [XIENCE™ Stent] patients enrolled in STOPDAPT.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT-3 Trial Design and Randomization11

- PCI with planned exclusive use of CoCr-EES (XIENCE)
- ACS presentation or ARC-HBR
- Eligible for DAPT (Aspirin/P2Y12inhibitor) for 1 month
Study design and Randomization
Group 1:
0 to 1-month: Aspirin + P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
Group 2:
0 to 1-month: P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
STOPDAPT-3 Trial 11 was designed to explore 0-month DAPT* (SAPT˄ using only P2Y12 inhibitor) for ACS and HBR patients.
Though the results are comparable for both bleeding and ischemic events for DAPT and SAPT arms, the study did not meet its endpoint and concluded to use DAPT for 1 month after PCI.
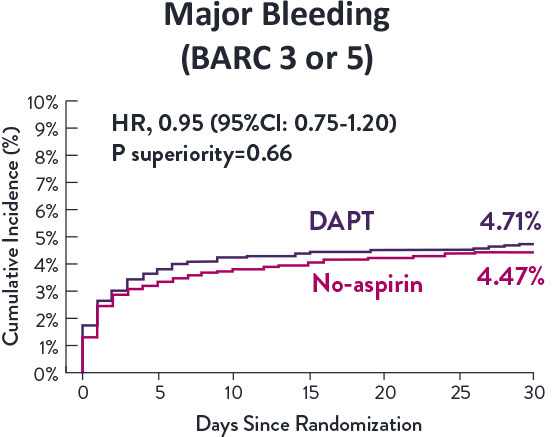
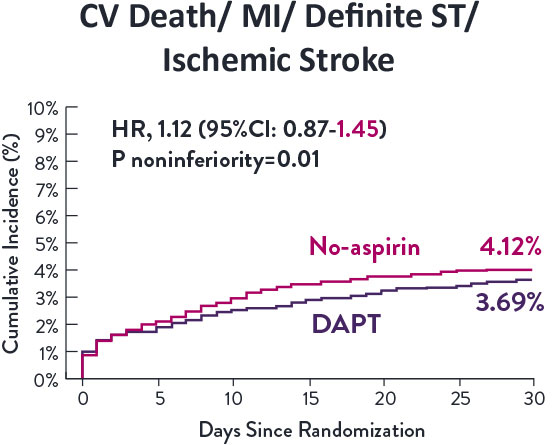

XIENCE™ Stent remains the ONLY DES with the shortest DAPT indication, as short as 28 days.12


Referencias
- Diamondback 360™ Coronary Orbital Atherectomy System Instructions for Use (IFU). Refer to IFU for additional information.
- Chambers, J., et al., JACC Cardiovasc Interv. 2014;7(5):510-518.
- Shlofmitz, E., et al., Expert Rev Med Devices. 2017;14(11):867-879.
- Yamamoto, M., et al., Catheter Cardiovasc Interv. 2019;93(7):1211-1218.
- Kini, A., et al., Catheter Cardiovasc Interv. 2015;86(6):1024-1032.
- Lee, MS, et al., J Invasive Cardiol. 2016;28(4):147-150.
- Moses, J., Cardiac Interventions Today. 2015;May/June:75. Link accessed November 20, 2023. https://citoday.com/articles/2015-may-june/vessel-preparation-with-orbital-atherectomy-for-treating-severe-coronary-calcium-for-stent-delivery.
- Data on file at Abbott.

MAT-XXXXXXX



