

La aterectomía orbital es un enfoque de primera línea para la preparación ideal del vaso en casos complejos de angioplastia con calcio severo.
Desafíos asociados con el calcio4
- Propenso a disección durante la angioplastia con balón o pre-dilatación
- Difícil de dilatar completamente el balón
- Puede impedir la entrega del stent
- Puede impedir la expansión y aposición adecuadas del stent
El Diamondback 360™ OAS ha demostrado ser eficaz y seguro en el tratamiento de lesiones severamente calcificadas, estudiado en más de 2,200 pacientes en 11 estudios robustos.5,6
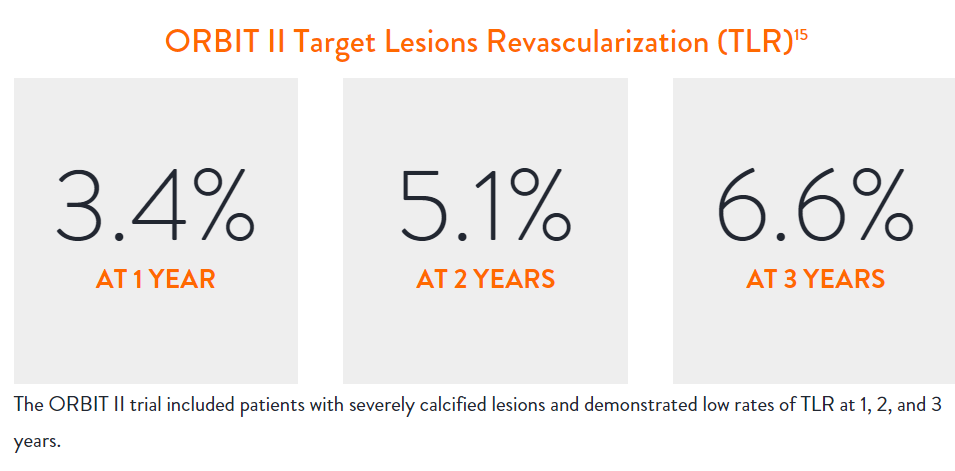
Estudio Clínico ORBIT II7
El estudio principal ORBIT II fue un estudio prospectivo y multicéntrico realizado en EE. UU. que evaluó a pacientes con lesiones coronarias gravemente calcificadas tratados con el Sistema de Aterectomía Orbital Coronaria Diamondback 360™ (OAS) Corona Clásica antes de la implantación del stent.
Principales Conclusiones
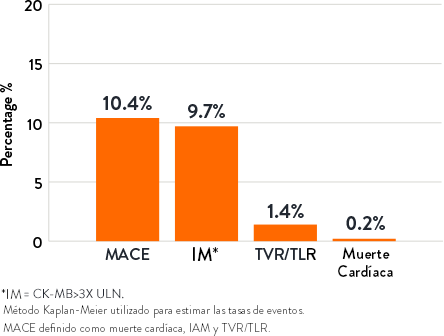
- El ORBIT II alcanzó los objetivos primarios de seguridad y eficacia por un margen significativo7
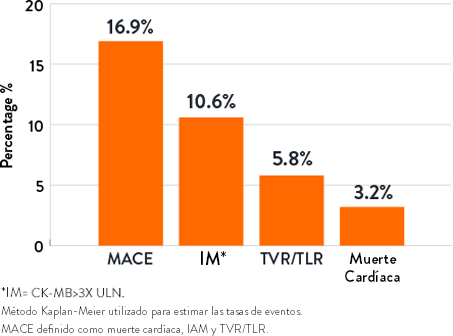
- El ORBIT II demostró resultados duraderos a largo plazo con una baja tasa de revascularización de la lesión objetivo (TLR) de 3,4% en 1 año y 6,6% en 3 años en los subconjuntos de stents farmacológicos (N=389/443)8
Diseño del estudio7
- 443 pacientes con lesiones severamente calcificadas fueron inscritos en 49 centros en EE. UU
- Diseñado para evaluar la seguridad y eficacia de la corona Diamondback 360™ Coronary OAS Classic
- Resultado primario de seguridad: Eventos cardíacos adversos mayores (MACE) en 30 días
- Resultado primario de eficacia: Éxito del procedimiento definido como éxito en la facilitación de la entrega del stent con estenosis residual de <50% y sin la ocurrencia de MACE intrahospitalario
POBLACIÓN DE PACIENTES7
| N=443 | |
|---|---|
| Historial de diabetes | 36.1% |
| Historial de CABG | 14.7% |
| Historial de dislipidemia | 91.9% |
| Historial de hipertensión | 91.6% |
| Fumador (fumador o exfumador) | 66.1% |
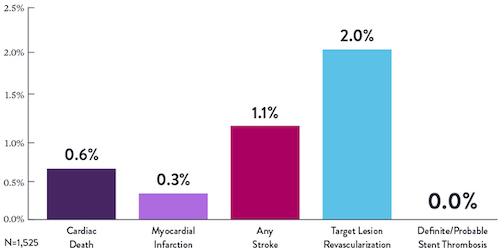
RESULTADO DEL ESTUDIO7
| Éxito del procedimiento | 88.9% |
|---|---|
| Entrega exitosa del stent | 97.7% |
| Menos del 50% de estenosis residual | 98.6% |
| Libertad de MACE intrahospitalario | 90.2% |
|
Resultados de 30 días9 |
Resultados a 1 año9 |
 |
 |

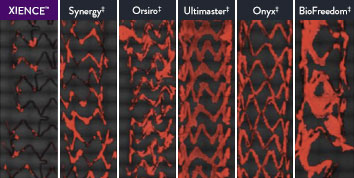
The XIENCE™ Stent is also recognized as being significantly more anti-thrombotic than other DES on the market. As shown in the study findings, XIENCE™ Stent reveals significantly less (p < 0.01) platelet adhesion—shown in red in the confocal microscopy images—than other DES, and platelet adhesion is an important factor in stent thrombosis.*8 These findings suggest that this stent choice “may be ideally suited for very short-term DAPT.”8
*Ex Vivo Swine Shunt Model.

Diamondback 360™ OAS Gives You the Versatility to Treat Challenging Cases
Treat even the most severely calcified lesions, with under 2-minute setup7,8 and predictable procedure times.2
Facilitates antegrade and retrograde treatment of:
- Long, Diffuse Lesions
Successfully treated lesions up to 60 mm in length in real-world study.9 - Heavily Stenosed Lesions
Crossed >99% of lesions with <2% pre-dilatation in the ORBIT II study.1,10 - Nodular Lesions
Effectively treats nodular calcification.5,6 - Ostial Lesions
Safely treats ostial lesions.11-13
Low Profile
6F compatible for femoral or radial access.14
Multiple Vessel Sizes
A single 1.25 mm crown treats vessels 2.5 mm to 4.0mm14
|
|
Proven Safety |
Procedural Success |
Low Q-Wave MI Rate |
>2,200 |
<1% |
97.7% |
0.9% |
|
Patients Across 11 |
Component Angiographic |
Crossing and Stent |
In the ORBIT II |
Sustained Clinical Performance
Data are for ORBIT II TLR in the OA+DES patient cohort.

“Stopping DAPT at 3 months in selected patients after [XIENCE™ Stent] implantation was at least as safe as the prolonged DAPT regimen adopted in the historical control group.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT 2 Trial Design and Randomization10

Short 1-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- After 1-month: Clopidogrel monotherapy

12-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- 1 to 12-month: Aspirin + Clopidogrel
- 12 to 60-month: Aspirin monotherapy

- Successful PCI using CoCr everolimus-eluting stent: XIENCE™
- Eligible for DAPT (aspirin/P2Y12 receptor blocker) for 1 year

- Patients who need oral anticoagulants
- History of intracranial bleeding
- Major in-hospital complications (MI/stroke/major bleeding)
STOPDAPT Study: XIENCE™ Stent with 3-Month DAPT Is Feasible9
STOPDAPT9 was the first prospective trial to study DAPT cessation at 3 months after implantation. Among other 1-year outcomes, the XIENCE™ Stent rate of stent thrombosis was 0.0%.
STOPDAPT Study Demonstrates Feasibility of XIENCE™ Stent with 3-Month DAPT9

Learn more about STOPDAPT 2

“It was noteworthy that no definite or probable stent thrombosis occurred in [XIENCE™ Stent] patients enrolled in STOPDAPT.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT-3 Trial Design and Randomization11

- PCI with planned exclusive use of CoCr-EES (XIENCE)
- ACS presentation or ARC-HBR
- Eligible for DAPT (Aspirin/P2Y12inhibitor) for 1 month
Study design and Randomization
Group 1:
0 to 1-month: Aspirin + P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
Group 2:
0 to 1-month: P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
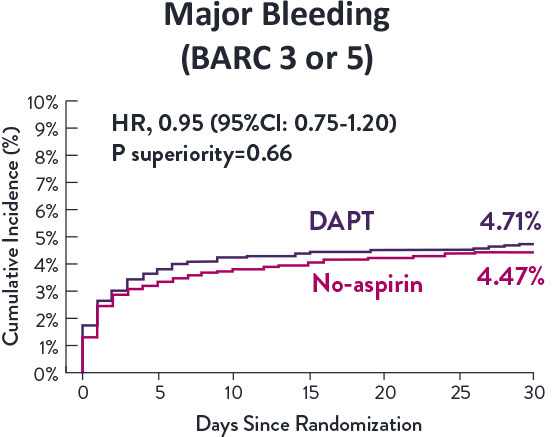
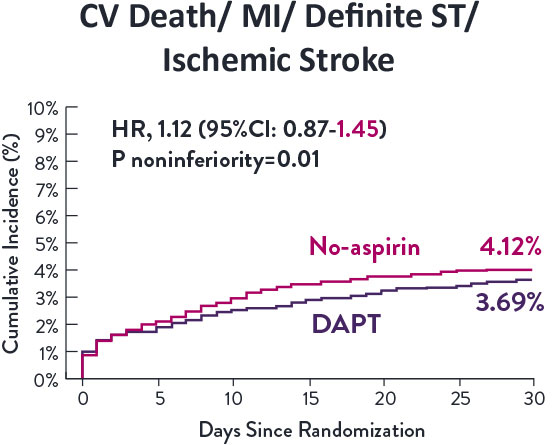
STOPDAPT-3 Trial 11 was designed to explore 0-month DAPT* (SAPT˄ using only P2Y12 inhibitor) for ACS and HBR patients.
Though the results are comparable for both bleeding and ischemic events for DAPT and SAPT arms, the study did not meet its endpoint and concluded to use DAPT for 1 month after PCI.

XIENCE™ Stent remains the ONLY DES with the shortest DAPT indication, as short as 28 days.12


Referencias
- Mintz GS. Intravascular imaging of coronary calcification and its clinical implications. JACC Cardiovasc Imaging. 2015;8(4):461-471.
- Mintz GS, et al. Patterns of calcification in coronary artery disease. A statistical analysis of intravascular ultrasound and coronary angiography in 1155 lesions. Circulation. 1995;91(7):1959-1965.
- Genereux P, et al. Ischemic outcomes after coronary intervention of calcified vessels in acute coronary syndromes. Pooled analysis from the HORIZONS-AMI (Harmonizing Outcomes With Revascularization and Stents in Acute Myocardial Infarction) and ACUITY (Acute Catheterization and Urgent Intervention Triage Strategy) TRIALS. J Am Coll Cardiol. 2014;63(18):1845-1854.
- Shlofmitz E, et al. Orbital atherectomy for the treatment of severely calcified coronary lesions: evidence, technique, and best practices. Expert Rev Med Devices. 2017;14(11):867-879.
- Shlofmitz, E., et al., Lesion Preparation with Orbital Atherectomy. Interv Cardiol. 2019;14(3):169-173. .
- Beohar, N., Orbital Atherectomy for Treating De Novo Severely Calcified Coronary Lesions. A Tertiary Center Experience, Presented at TCT Connect 2020.
- Chambers JW, et al. Pivotal trial to evaluate the safety and efficacy of the orbital atherectomy system in treating de novo, severely calcified coronary lesions (ORBIT II). JACC Cardiovasc Interv. 2014;7(5):510-518.
- Data on file at Abbott.
- Lee M, et al. Orbital atherectomy for treating de novo, severely calcified coronary lesions: 3-year results of the pivotal ORBIT II trial. Cardiovasc Revasc Med. 2017;18(4):261-264.

MAT-XXXXXXX



