

Alguns removem o cálcio. Outros fraturam. O Sistema de Aterectomia Orbital Periférica Stealth 360™ faz as duas coisas com um único dispositivo.
O Sistema de Aterectomia Orbital Periférica Stealth 360™ remove com segurança o cálcio intimal e fratura o cálcio medial com seu exclusivo mecanismo de dupla ação, oferecendo uma solução abrangente para o cálcio - tudo com um único dispositivo.1,4
Remove o Cálcio Intimal + Fratura o Cálcio Medial
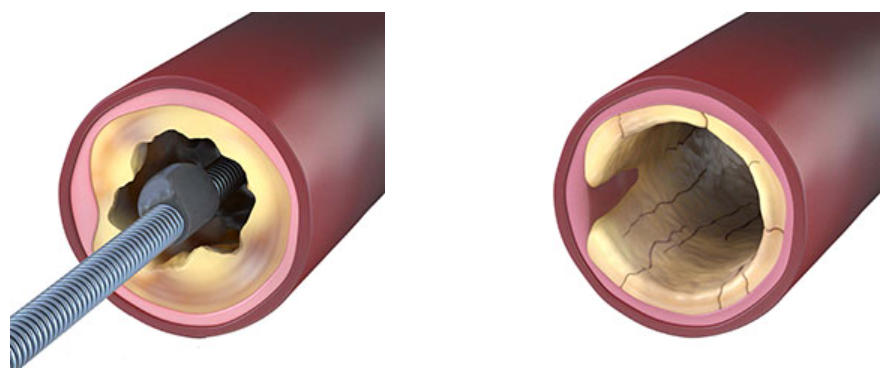
Assista ao vídeo para ver como funciona.
O Sistema de Aterectomia Orbital Periférica Stealth 360™ possui resultados de longo prazo, com segurança comprovada e alta versatilidade.
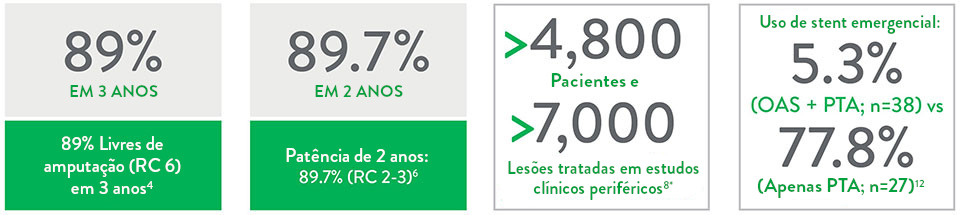
Eficácia de longo prazo:
- 100% livres de amputação maior em 3 anos em RC 2-3 (95% em RC 4-5, 89% em RC 6)4
- 82,1% de patência em 1 ano (RC 2-3)5
- 89,7% de patência em 2 anos (RC 2-3)6
- Cicatrização significativa das feridas no grupo CLTI (RC 4-6) em 2 anos6
Registro comprovado de segurança:
- Excelente segurança no procedimento7-11
- O uso de aterectomia orbital + PTA de baixa pressão resultou em baixa taxa de eventos adversos maiores10 e menor uso de stents emergenciais12
Uso versátil:
- Uma única coroa pode ser usada em vasos acima e abaixo do joelho e em diferentes tipos de calcificação e lesões1,2,9,10,12
- Moderada a severa
- Macia/dura
- Curta/longa
- Excêntrica
- Concêntrica
- Moderada a severa
- O design de baixo perfil pode ajudar a minimizar as complicações do local de acesso13 e melhorar o alcance do procedimento4
- Prepara vasos menores e maiores para o tratamento sem a necessidade de aumentar o tamanho do cateter e da bainha1,2

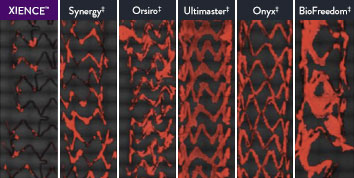
The XIENCE™ Stent is also recognized as being significantly more anti-thrombotic than other DES on the market. As shown in the study findings, XIENCE™ Stent reveals significantly less (p < 0.01) platelet adhesion—shown in red in the confocal microscopy images—than other DES, and platelet adhesion is an important factor in stent thrombosis.*8 These findings suggest that this stent choice “may be ideally suited for very short-term DAPT.”8
*Ex Vivo Swine Shunt Model.

Diamondback 360™ OAS Gives You the Versatility to Treat Challenging Cases
Treat even the most severely calcified lesions, with under 2-minute setup7,8 and predictable procedure times.2
Facilitates antegrade and retrograde treatment of:
- Long, Diffuse Lesions
Successfully treated lesions up to 60 mm in length in real-world study.9 - Heavily Stenosed Lesions
Crossed >99% of lesions with <2% pre-dilatation in the ORBIT II study.1,10 - Nodular Lesions
Effectively treats nodular calcification.5,6 - Ostial Lesions
Safely treats ostial lesions.11-13
Low Profile
6F compatible for femoral or radial access.14
Multiple Vessel Sizes
A single 1.25 mm crown treats vessels 2.5 mm to 4.0mm14
|
|
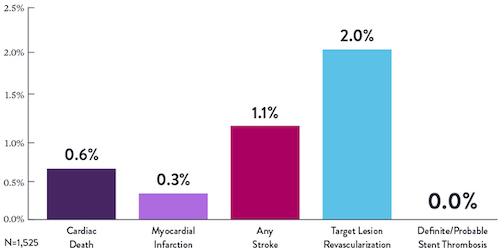
Proven Safety |
Procedural Success |
Low Q-Wave MI Rate |
>2,200 |
<1% |
97.7% |
0.9% |
|
Patients Across 11 |
Component Angiographic |
Crossing and Stent |
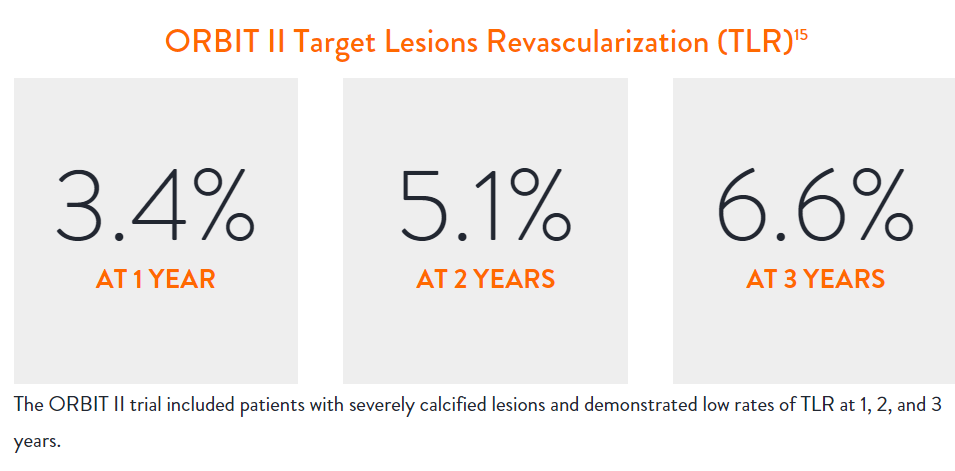
In the ORBIT II |
Sustained Clinical Performance
Data are for ORBIT II TLR in the OA+DES patient cohort.

“Stopping DAPT at 3 months in selected patients after [XIENCE™ Stent] implantation was at least as safe as the prolonged DAPT regimen adopted in the historical control group.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT 2 Trial Design and Randomization10

Short 1-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- After 1-month: Clopidogrel monotherapy

12-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- 1 to 12-month: Aspirin + Clopidogrel
- 12 to 60-month: Aspirin monotherapy

- Successful PCI using CoCr everolimus-eluting stent: XIENCE™
- Eligible for DAPT (aspirin/P2Y12 receptor blocker) for 1 year

- Patients who need oral anticoagulants
- History of intracranial bleeding
- Major in-hospital complications (MI/stroke/major bleeding)
STOPDAPT Study: XIENCE™ Stent with 3-Month DAPT Is Feasible9
STOPDAPT9 was the first prospective trial to study DAPT cessation at 3 months after implantation. Among other 1-year outcomes, the XIENCE™ Stent rate of stent thrombosis was 0.0%.
STOPDAPT Study Demonstrates Feasibility of XIENCE™ Stent with 3-Month DAPT9

Learn more about STOPDAPT 2

“It was noteworthy that no definite or probable stent thrombosis occurred in [XIENCE™ Stent] patients enrolled in STOPDAPT.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT-3 Trial Design and Randomization11

- PCI with planned exclusive use of CoCr-EES (XIENCE)
- ACS presentation or ARC-HBR
- Eligible for DAPT (Aspirin/P2Y12inhibitor) for 1 month
Study design and Randomization
Group 1:
0 to 1-month: Aspirin + P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
Group 2:
0 to 1-month: P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
STOPDAPT-3 Trial 11 was designed to explore 0-month DAPT* (SAPT˄ using only P2Y12 inhibitor) for ACS and HBR patients.
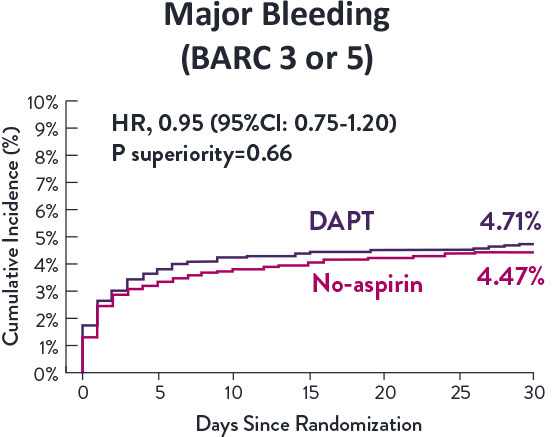
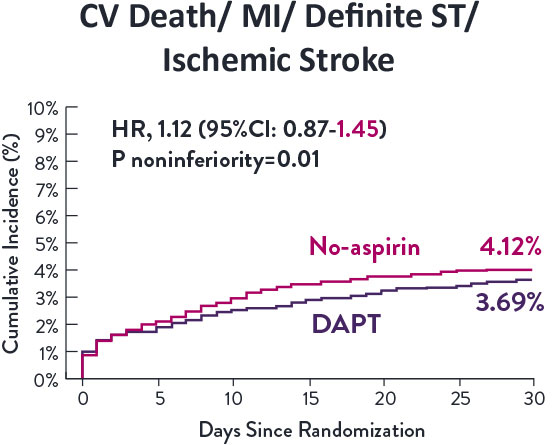
Though the results are comparable for both bleeding and ischemic events for DAPT and SAPT arms, the study did not meet its endpoint and concluded to use DAPT for 1 month after PCI.

XIENCE™ Stent remains the ONLY DES with the shortest DAPT indication, as short as 28 days.12


Referências
*Contagens atualizadas em 25 de março de 2020 – sujeitas a alterações (inclui PAD 1, PAD li, OASIS, CONFIRM, CALCIUM 360, COMPLIANCE 360, TRUTH, CLARITY1, LIBERTY360, OPTIMIZE BTK, & REACH PVI)
- Adams GL, et al. J Cardiovasc Transl Res. 2011;4(2):220-229.
- Mustapha JA, et al. J Crit Limb Ischem. 2021;1(3):E118-E125.
- Dados nos arquivos da Abbott. Clinical Report Memo on Peripheral and Coronary OAS Pulsatile Force and Calcium Fracture. 2021.
- Giannopoulos S, et al. J Endovasc Ther. 2020;27(5):714-725.
- Mustapha JA. Late Breaking: LIBERTY 360 Trial 2-Year Update. Apresentado no AMP 2018.
- Adams G, et al. J Vasc Surg. 2019;70(5, Suppl):e188-e189.
- Martinsen B, Evaluation and Use of Atherectomy Devices for CLI in US, Japan, and EU: Industry View. Apresentado no VIVA 2017 (Inclui laser, rotacional, direcional).
- Dados nos arquivos da Abbott.
- Das T, et al. Catheter Cardiovasc Interv. 2014;83:115-122. (Dissecções limitantes de fluxo e embolizações não foram acompanhadas em 1146 lesões)
- Shammas NW, et al. J Endovasc Ther. 2012;19:480-488.
- Babaev A, et al. Vasc Endovascular Surg. 2015;49:188-194.
- Dattilo R, et al. J Invasive Cardiol. 2014;26(8):355-360.
- Bosiers M, et al. J Endovasc Ther. 2013;20(6):746-756.

MAT-XXXXXXX



