

Uma abordagem especializada para DAP (Doença Arterial Periférica)
O Sistema de Aterectomia Orbital Periférica Stealth 360™ oferece ferramentas para abordar vários cenários de calcificação, vantagens de baixo perfil e múltiplas soluções de acesso.
Conheça o Dispositivo Stealth 360™

Opções de Coroas Stealth 360™
Coroas revestidas de diamante montadas excentricamente com capacidades bidirecionais tratam uma ampla gama de vasos, permitindo o tratamento de múltiplas lesões e tamanhos de artérias com um único dispositivo.

Coroa Micro
- Projetada com flexibilidade para vasos tortuosos e curvas fechadas abaixo do joelho1
- O modo GlideAssist™ para a Coroa Micro de 1,25 permite que a coroa rotacione em baixa velocidade (5k RPM) para facilitar a navegabilidade e a remoção do dispositivo sobre o fio-guia¹

Coroa Sólida
- Projetada para lesões calcificadas moderadas a severas1
- Oferece lixamento eficiente em calcificação2
- A superfície revestida com diamante permite tempos de execução curtos1

Coroa Clássica
- A mais flexível das nossas configurações de coroa1
- Projetada para vasos tortuosos, lesões ostiais e procedimentos distais abaixo do joelho1
Componentes do Sistema Stealth 360™
Mecanismo de bombeamento para aterectomiaA bomba é montada diretamente em um suporte para soro e fornece energia e o mecanismo de bombeamento para o Sistema Stealth 360™. |
 |
Lubrificante para dispositivo de aterectomia ViperSlide™
ViperSlide™ é um lubrificante exclusivo para uso com os Sistemas de Aterectomia Orbital da Abbott. O ViperSlide™ aumenta a lubrificação, reduzindo assim o atrito entre o dispositivo de Aterectomia e o Fio-Guia ViperWire Advance™. |
 |
Fios-guia periféricos ViperWire Advance™ e ViperWire Advance™ com ponta flexível
Os fios-guia periféricos ViperWire Advance™ e ViperWire Advance™ com ponta flexível foram projetados especificamente para os Sistemas de Aterectomia Orbital Periférica Stealth 360™ para facilitar o posicionamento adequado do dispositivo dentro da artéria periférica e permitir ganho orbital durante o tratamento da DAP. |
 |

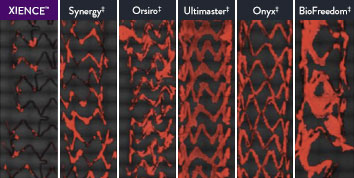
The XIENCE™ Stent is also recognized as being significantly more anti-thrombotic than other DES on the market. As shown in the study findings, XIENCE™ Stent reveals significantly less (p < 0.01) platelet adhesion—shown in red in the confocal microscopy images—than other DES, and platelet adhesion is an important factor in stent thrombosis.*8 These findings suggest that this stent choice “may be ideally suited for very short-term DAPT.”8
*Ex Vivo Swine Shunt Model.

Diamondback 360™ OAS Gives You the Versatility to Treat Challenging Cases
Treat even the most severely calcified lesions, with under 2-minute setup7,8 and predictable procedure times.2
Facilitates antegrade and retrograde treatment of:
- Long, Diffuse Lesions
Successfully treated lesions up to 60 mm in length in real-world study.9 - Heavily Stenosed Lesions
Crossed >99% of lesions with <2% pre-dilatation in the ORBIT II study.1,10 - Nodular Lesions
Effectively treats nodular calcification.5,6 - Ostial Lesions
Safely treats ostial lesions.11-13
Low Profile
6F compatible for femoral or radial access.14
Multiple Vessel Sizes
A single 1.25 mm crown treats vessels 2.5 mm to 4.0mm14
|
|
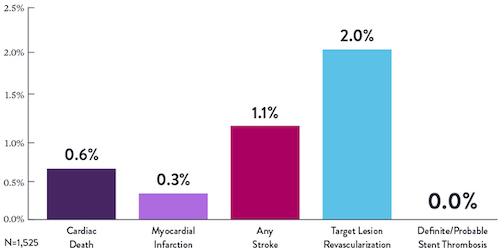
Proven Safety |
Procedural Success |
Low Q-Wave MI Rate |
>2,200 |
<1% |
97.7% |
0.9% |
|
Patients Across 11 |
Component Angiographic |
Crossing and Stent |
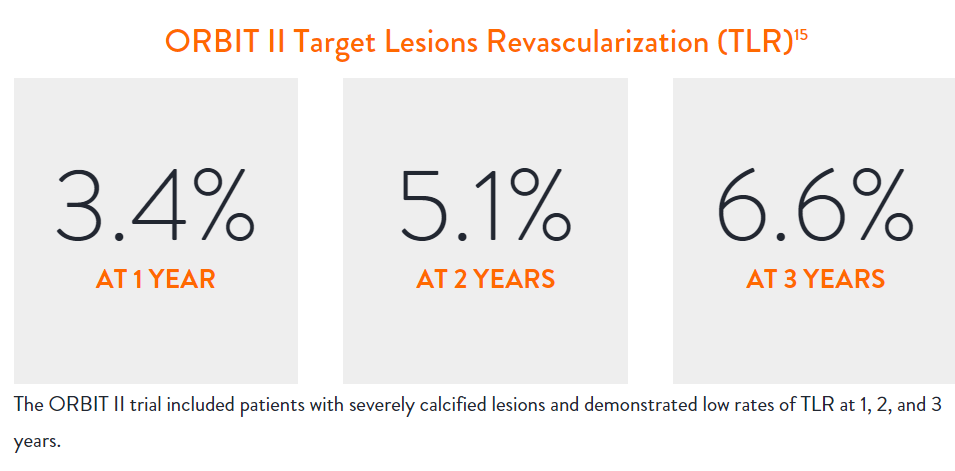
In the ORBIT II |
Sustained Clinical Performance
Data are for ORBIT II TLR in the OA+DES patient cohort.

“Stopping DAPT at 3 months in selected patients after [XIENCE™ Stent] implantation was at least as safe as the prolonged DAPT regimen adopted in the historical control group.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT 2 Trial Design and Randomization10

Short 1-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- After 1-month: Clopidogrel monotherapy

12-Month DAPT
- 0 to 1-month: Aspirin + P2Y12
- 1 to 12-month: Aspirin + Clopidogrel
- 12 to 60-month: Aspirin monotherapy

- Successful PCI using CoCr everolimus-eluting stent: XIENCE™
- Eligible for DAPT (aspirin/P2Y12 receptor blocker) for 1 year

- Patients who need oral anticoagulants
- History of intracranial bleeding
- Major in-hospital complications (MI/stroke/major bleeding)
STOPDAPT Study: XIENCE™ Stent with 3-Month DAPT Is Feasible9
STOPDAPT9 was the first prospective trial to study DAPT cessation at 3 months after implantation. Among other 1-year outcomes, the XIENCE™ Stent rate of stent thrombosis was 0.0%.
STOPDAPT Study Demonstrates Feasibility of XIENCE™ Stent with 3-Month DAPT9

Learn more about STOPDAPT 2

“It was noteworthy that no definite or probable stent thrombosis occurred in [XIENCE™ Stent] patients enrolled in STOPDAPT.”
— Masahiro Natsuaki, MD, STOPDAPT Trial9

STOPDAPT-3 Trial Design and Randomization11

- PCI with planned exclusive use of CoCr-EES (XIENCE)
- ACS presentation or ARC-HBR
- Eligible for DAPT (Aspirin/P2Y12inhibitor) for 1 month
Study design and Randomization
Group 1:
0 to 1-month: Aspirin + P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
Group 2:
0 to 1-month: P2Y12 (Prasugrel)
After 1-month: Clopidogrel monotherapy
STOPDAPT-3 Trial 11 was designed to explore 0-month DAPT* (SAPT˄ using only P2Y12 inhibitor) for ACS and HBR patients.
Though the results are comparable for both bleeding and ischemic events for DAPT and SAPT arms, the study did not meet its endpoint and concluded to use DAPT for 1 month after PCI.
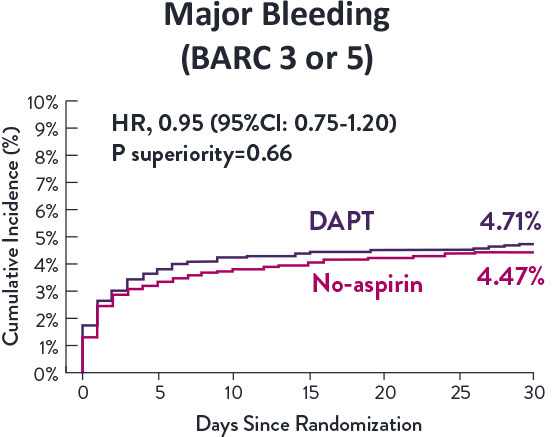
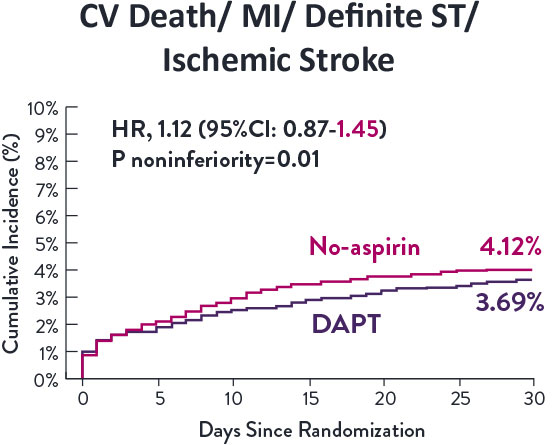

XIENCE™ Stent remains the ONLY DES with the shortest DAPT indication, as short as 28 days.12


Referências
- Dados arquivados na Abbott.
- Adams GL, et al. Optimal Techniques with the Diamondback 360 System Achieve Effective Results for the Treatment of Peripheral Artery Disease. J Cardiovasc Transl Res. 2011 Apr; 4(2):220-9.

MAT-XXXXXXX



